
Mostly Reactive Except for the noble ones. Atoms are not independently in nature, but molecules can be.Atoms are constituents of matter, while molecules are made up of atoms different from each other.Key differences between Atom and Molecule This molecular formula is nothing more than the name of the element, followed by the number of atoms of that element in the molecule. Generally, molecules are represented in a molecular formula. While in the solid-state, the molecules are tighter or close together and can only vibrate. In the liquid state, the molecules tend to be loosely grouped the reason why they can move. For example, in a gaseous state, molecules usually fly around. Because of this, the molecules can have hexagonal, pentagonal forms.įinally, molecules tend to cluster and their formation depends on their state. The atoms of the molecules are adhered to each other in a particular way, depending on the number of bonds that the atom can make. When molecules are formed, they can exist independently in nature so we can always find a molecule of oxygen, carbon, or water. On the other hand, a single atom cannot exist independently in nature (without joining something).įor example, we will never find a single atom of oxygen, it is always attached to something (oxygen = O2). The molecules can have properties very different from those that have the elements that compose them.įor example, water behaves very differently from oxygen and hydrogen. However, such atoms cannot be separated further without provoking a nuclear reaction. It is constituted by atoms that are different from each other and for this reason, each of them can be separated. This happens through chemical reactions.įor example, two atoms of hydrogen and one of oxygen, combine to make a molecule of water.Ī molecule is the smallest amount that can exist in a substance. The atoms of different elements unite and form molecules. On the other hand, neutrons have no electric charge. 
The electrons have a negative electric charge and this is why they are attracted to the nucleus since the protons have a positive electric charge. While the electrons that spin around, have attached to the nucleus thanks to the electromagnetic force. Protons and neutrons are found in the nucleus, located in the center of atoms. The atoms of each element are composed of protons, neutrons, and electrons. To date, 118 elements have been discovered and have been divided into metals, metalloids, and nonmetals.Įach element has its own set of properties and most are found on earth while a few have developed from nuclear reactions.Īll elements can be found in the periodic table, listed according to their atomic number. The atomic number derives from the number of protons present in the nucleus of the atoms of the element. There are many types of atoms and each has a unique number, mass, and size.Ĭlusters of atoms of the same type constitute the chemical elements.Īn element is a pure chemical substance that has atoms of the same class and we distinguish it by its atomic number. The elements which occur in naturally on earth are called natural elements.They are so small that we cannot see them through even via a common microscope. Mosley is called the father of modern periodic table. The 8 most abundant elements in Earth’s crust (by mass) are the following :ĭmitri Ivanovich Mendeleev is the father of periodic table. The first detailed form of the periodic table was developed by Mendeleev (based on mass number) but later Henry Gwyn Jeffrey’s Moseley made a new periodic table based on atomic number.Oxygen is the most common element in the earth’s crust.Iron is the most abundant element (by mass) in the earth.Hydrogen and Helium are the most abundant elements in the universe.Carbon, Oxygen, Silicon, Arsenic, Aluminum, Iron, Copper, Gold, Mercury etc. ElementsĪ chemical element is a pure chemical substance consisting of one type of atom distinguished by its atomic number. Some molecules are made only by one type of atoms (two oxygen atoms bond together to form O2 molecule) while molecules like protein are made up of atoms from different elements. 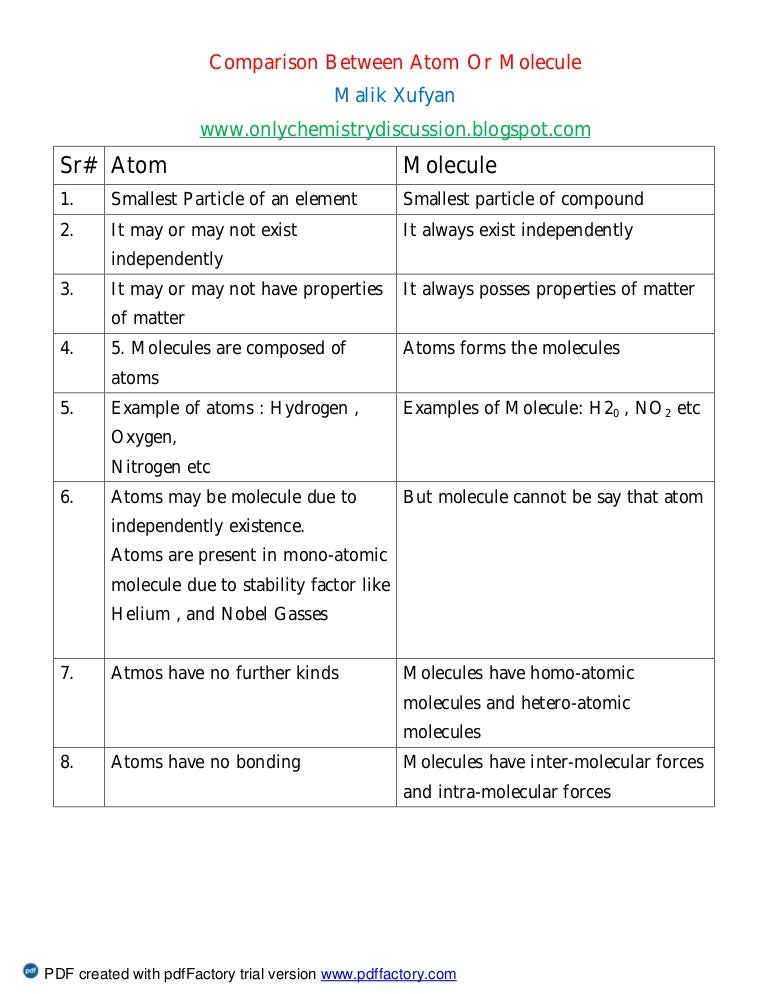
Molecules are made of atoms of one or more elements. Due to the presence of protons nucleus has a positive charge. Particles present inside the nucleus are called nucleons and they include mainly protons and neutrons. The central part of an atom is called nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
